When semaglutide first hit the market as a diabetes drug, nobody predicted it would end up in clinical trials for Alzheimer's disease, opioid addiction, and liver failure. Yet here we are in 2026, watching one of the most prescribed drug classes in history rewrite what we thought we knew about treating chronic disease. More than one in five U.S. adults have now used a GLP-1 receptor agonist, and the research catching up to that massive adoption curve keeps delivering surprises that have nothing to do with the scale.
Over the past 18 months, large-scale studies have linked GLP-1 drugs to reduced risk of dementia, lower rates of substance use disorders across five different drug categories, measurable improvements in serious liver disease, and protection against kidney failure. Some of these findings come from observational data and need further validation. Others come from rigorous randomized controlled trials that have already changed FDA-approved indications. Together, they paint a picture of a drug class whose full therapeutic potential is still being mapped.
Here is where the science actually stands, what the strongest evidence supports, where the gaps remain, and what all of it means if you or someone you know is taking these medications.
The Heart Benefits Are Real, and Getting Clearer
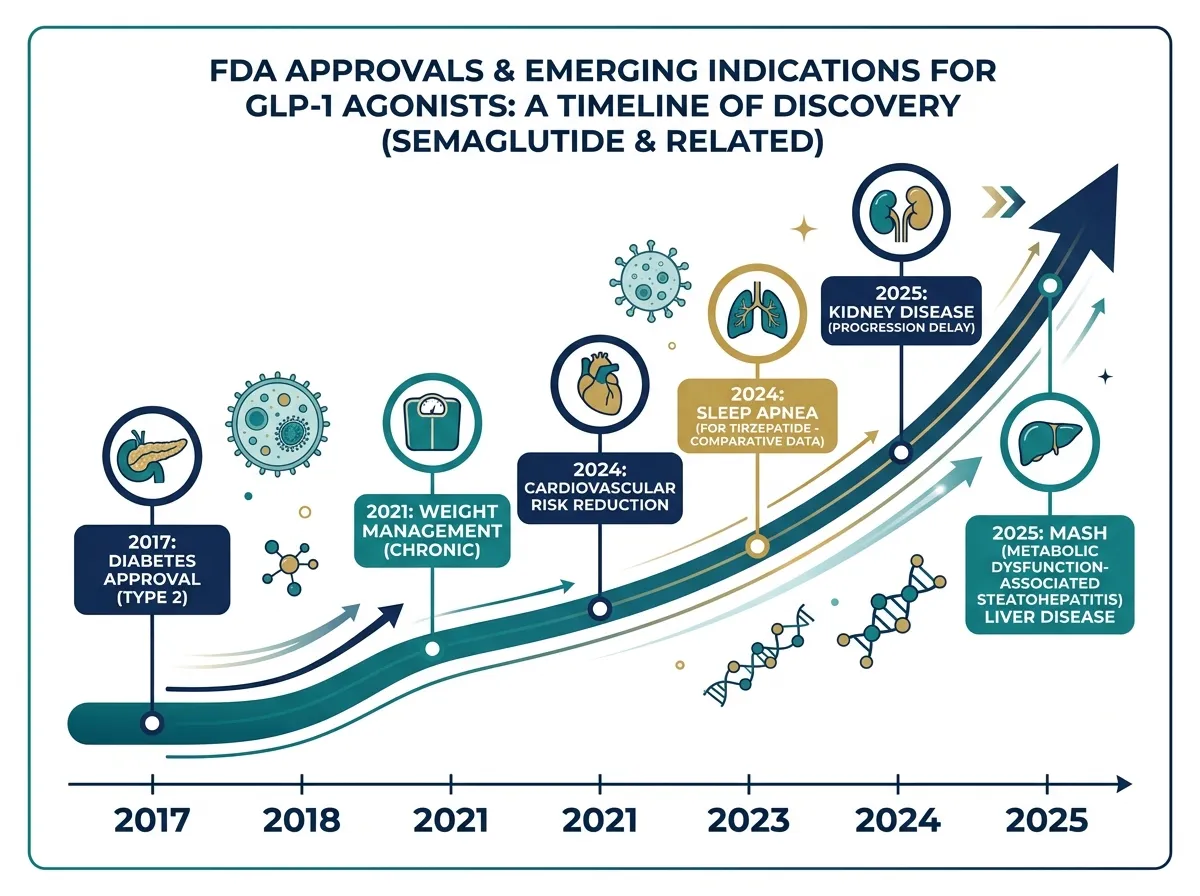
The cardiovascular story for GLP-1 drugs has been building since the SELECT trial, published in the New England Journal of Medicine in November 2023. That study enrolled 17,604 overweight or obese adults with existing cardiovascular disease and found that semaglutide reduced major adverse cardiovascular events (heart attack, stroke, or cardiovascular death) by 20% compared to placebo. The FDA expanded semaglutide's approved uses to include cardiovascular risk reduction in March 2024.
But the more recent data go further. Muthiah Vaduganathan, a cardiologist at Brigham and Women's Hospital and Harvard Medical School, along with colleague Nils Kruger, found that GLP-1 drugs achieved a 40% relative risk reduction in heart failure with preserved ejection fraction compared to older diabetes medications. That specific type of heart failure, where the heart muscle stiffens and can't fill properly, has been notoriously difficult to treat. For years, cardiologists had limited options. The fact that a class of drugs originally designed to manage blood sugar is outperforming dedicated cardiac therapies in this area has shifted how researchers think about the relationship between metabolic health and heart function.
Josephine Li, clinical director of the Diabetes Center at Massachusetts General Hospital and Harvard Medical School, notes that the drugs "reduce the risk of major adverse cardiovascular events like cardiovascular death, nonfatal myocardial infarction, nonfatal stroke." The mechanism likely involves more than just weight loss. GLP-1 receptor agonists simultaneously lower blood pressure, improve cholesterol profiles, reduce inflammatory markers, and enhance insulin sensitivity. Each of those effects independently reduces cardiovascular risk, and their combination may explain why the heart benefits appear stronger than weight loss alone would predict.
A 600,000-Person Study on Addiction, and the Results Are Striking
Perhaps the most unexpected line of GLP-1 research involves substance use disorders. In March 2026, the BMJ published a study led by Ziyad Al-Aly, a clinical epidemiologist and chief of research and development at the VA St. Louis Health Care System, analyzing electronic health records from 606,434 U.S. veterans with type 2 diabetes over three years.
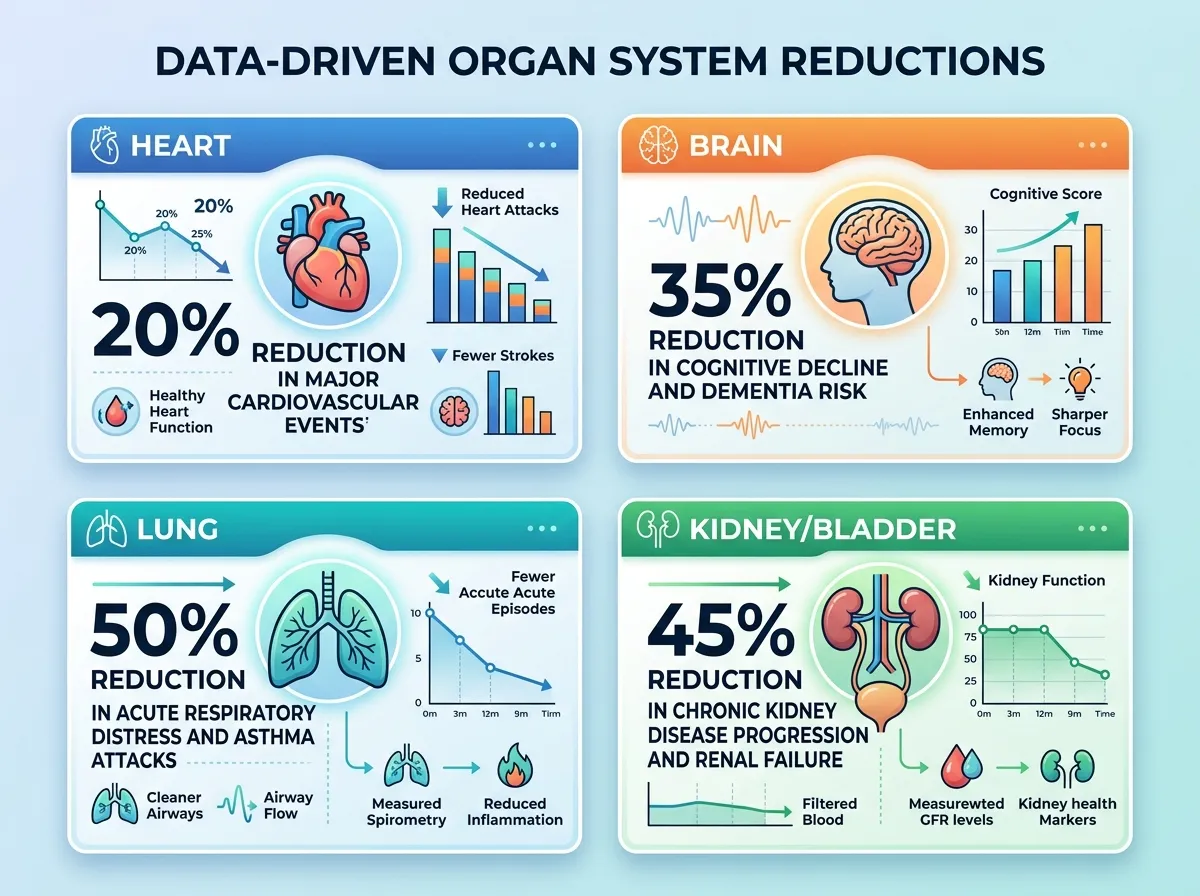
The findings were broad and consistent. Veterans taking GLP-1 drugs had a 14% lower risk of developing any substance use disorder compared to those on other diabetes medications. The reductions spanned every substance category the researchers examined: 18% lower risk for alcohol use disorders, 14% for cannabis, 20% for cocaine, 20% for nicotine, and 25% for opioid use disorders.
For veterans who already had a substance use disorder, the numbers were even more dramatic. GLP-1 users saw 31% fewer emergency department visits, a 26% decline in hospital admissions, a 39% decrease in overdoses, and a 50% reduction in drug-related deaths.
"This is an obesity and diabetes drug; this is not an addiction drug," Al-Aly told Scientific American. "So the big surprise was: it was consistently working across all substances."
The biological explanation centers on dopamine. GLP-1 receptors are expressed in brain regions that regulate reward and motivation, including the nucleus accumbens and ventral tegmental area. Addictive substances hijack these same circuits, flooding them with dopamine to create compulsive use patterns. GLP-1 drugs appear to modulate that reward signaling, not by blocking pleasure entirely, but by reducing the intensity of cravings. The same mechanism that quiets "food noise" for people trying to lose weight may be quieting the neurological pull of alcohol, nicotine, and other substances.
The study has limitations. The veteran population skews older, male, and white, though women in the dataset showed similar trends. The researchers did not compare different GLP-1 drug types or dosages. And observational studies, no matter how large, cannot prove causation the way randomized controlled trials can. Still, with over 15 clinical trials now underway globally to test GLP-1 drugs for various addictions, according to Harvard's Mary Shen, a resident physician at Brigham and Women's Hospital, the early signal is strong enough to justify serious investigation.
The Brain: A Complicated Picture
The cognitive health data for GLP-1 drugs tell a split story that illustrates why careful reading of research matters more than headlines.
On the observational side, the evidence looks promising. An analysis of more than 100 million U.S. patient records found that Ozempic use was associated with lower risk of cognitive problems. A separate study of 2.5 million Veterans Affairs patients found that GLP-1 users developed dementia 40 to 70% less often than people taking other diabetes medications. These are large datasets with consistent signals.
But when Novo Nordisk tested oral semaglutide directly in the EVOKE and EVOKE+ trials, enrolling over 3,800 participants with mild cognitive impairment or early-stage Alzheimer's, the drug failed to slow disease progression. The results, announced in late 2025, showed no significant benefit on cognition or function.
The contrast sharpened when a different GLP-1 drug produced better results in a smaller trial. Professor Paul Edison of Imperial College London led the ELAD study, which tested injectable liraglutide in 204 non-diabetic patients with mild to moderate Alzheimer's. Participants receiving liraglutide experienced nearly 50% less brain volume loss and 18% slower cognitive decline compared to placebo.
Edison offered an explanation for the divergent outcomes. The oral semaglutide used in EVOKE was chemically optimized for absorption through the gut, which may have limited how much of the drug actually reached the brain. Injectable liraglutide, while a smaller molecule with different pharmacokinetics, appears to penetrate the blood-brain barrier in small amounts. "The negative trial result may indicate lack of drug access to the brain, rather than failure of the concept itself," Edison noted.
The takeaway is nuanced. GLP-1 drugs probably won't treat established Alzheimer's disease, at least not in their current formulations. But they may help prevent dementia when taken earlier, potentially through their effects on inflammation, insulin signaling, and vascular health, all of which play roles in neurodegeneration. The distinction between prevention and treatment is critical, and future trials targeting people at high risk for dementia before symptoms appear could clarify whether the strong observational signal translates into a real clinical tool.
Liver Disease: The ESSENCE Trial Changes the Standard of Care
For patients with metabolic dysfunction-associated steatohepatitis (MASH, formerly known as NASH), the data from semaglutide are among the most clear-cut of any condition discussed in this article.
The ESSENCE trial, a Phase 3 multicenter randomized controlled study published in the New England Journal of Medicine in 2025, enrolled 1,197 patients with biopsy-confirmed MASH and stage 2 or 3 liver fibrosis. Patients received either weekly subcutaneous semaglutide at 2.4 mg or placebo for up to 240 weeks, with a planned interim analysis at 72 weeks involving the first 800 patients.
The results at that interim analysis were strong enough to change clinical practice. Resolution of liver inflammation without worsening fibrosis occurred in 62.9% of semaglutide patients versus 34.3% on placebo, a nearly two-to-one advantage. Improvement in liver fibrosis without worsening inflammation occurred in 36.8% of the semaglutide group versus 22.4% on placebo. And 32.7% of semaglutide patients achieved both outcomes simultaneously, compared to just 16.1% receiving placebo.
MASH affects an estimated 6 to 8 million Americans and can progress to cirrhosis, liver failure, and liver cancer. Before semaglutide, treatment options were limited largely to lifestyle modification and, for the most advanced cases, liver transplantation. The FDA approved semaglutide for MASH treatment in August 2025, giving clinicians a pharmacological tool for a disease that had very few effective interventions.
What makes the liver data particularly compelling is the biological logic behind it. MASH is fundamentally a metabolic disease driven by insulin resistance, excess fat accumulation in liver cells, and chronic inflammation. GLP-1 drugs address all three pathways simultaneously. They improve insulin sensitivity, reduce visceral and hepatic fat, and lower systemic inflammatory markers. The liver benefits aren't a mysterious side effect; they're a direct consequence of the drug's core metabolic mechanisms working on an organ that sits at the center of metabolic processing.
Kidney Protection: A Trial Stopped Early for Efficacy
The kidney data add another organ system to the growing list. The FLOW trial tested semaglutide in over 3,500 patients with type 2 diabetes and chronic kidney disease (CKD), and the trial was stopped early because the drug was performing well enough that continuing to give some patients placebo was considered unethical by the data safety monitoring board.
Semaglutide significantly reduced the risk of kidney complications in these patients, leading the FDA to approve Ozempic for this indication in January 2025. For the estimated 37 million Americans living with chronic kidney disease, many of whom also have type 2 diabetes, this adds a meaningful option to a treatment toolkit that has historically focused on blood pressure management and glucose control.
The kidney protection likely works through multiple mechanisms. Better blood sugar control reduces the glycemic damage that accelerates kidney deterioration. Lower blood pressure decreases the mechanical stress on delicate kidney filtration units. Reduced inflammation slows the fibrotic scarring that gradually destroys kidney tissue. And weight loss itself reduces the metabolic burden on kidneys already working under strain.
What Comes Next: The Research Pipeline
The list of conditions being actively investigated for GLP-1 treatment continues to expand. Researchers are running trials on inflammatory bowel disease, various forms of arthritis, polycystic ovary syndrome (PCOS), and even certain obesity-related cancers. A study of 17,000 obese adults found that GLP-1 users had a 19% lower risk of death from COVID-19 infection. Early research into type 1 diabetes, where these drugs are not currently approved, is also underway.
The biological rationale for this breadth of investigation comes back to where GLP-1 receptors actually live in the body. Unlike drugs designed to hit a single target in a single organ, GLP-1 receptors are expressed in the brain, heart, liver, kidneys, gut, pancreas, and immune cells. A drug that activates these receptors is, by nature, a systemic intervention. The question researchers are working to answer is not whether GLP-1 drugs affect multiple organ systems (they clearly do) but which of those effects are clinically meaningful enough to justify treatment and which are statistical noise in large datasets.
That distinction matters because the enthusiasm around GLP-1 drugs carries real risks. Off-label prescribing for conditions where the evidence is still preliminary exposes patients to side effects (nausea, vomiting, potential muscle loss, and rare but serious risks including pancreatitis and thyroid concerns) without proven benefit. The gap between "associated with lower risk in an observational study" and "proven effective in a randomized controlled trial" is enormous, and patients deserve clarity about which side of that gap any given claim falls on.
The research also needs to broaden its demographic reach. Most large GLP-1 trials have enrolled primarily white participants in North America and Europe. Genetic variation, dietary patterns, and comorbidity profiles differ across populations, and benefits observed in one group may not translate identically to others. The veteran study on substance use disorders, while large, skewed heavily toward older white men. Independent, publicly funded research that includes diverse populations is essential for understanding the full picture.
Your Action Plan
The accumulating evidence suggests that GLP-1 drugs are far more than weight loss medications. They are metabolic modulators with documented effects on the heart, liver, kidneys, and potentially the brain and addiction circuits. For the millions of people already taking these drugs, the expanding benefit profile is encouraging. For the medical community, it signals a need to rethink how these medications are categorized and prescribed.
A few practical points worth keeping in mind:
If you're currently taking a GLP-1 drug, the cardiovascular, liver, and kidney data suggest benefits you may already be receiving beyond weight management. Discuss these with your prescribing physician, particularly if you have risk factors for any of the conditions where evidence is strongest. Make sure you're also addressing nutrition while on GLP-1 therapy, since adequate protein and micronutrient intake remain critical regardless of which benefits you're aiming for.
If you're considering a GLP-1 drug, understand that the strongest evidence exists for weight loss, cardiovascular risk reduction (in people with existing heart disease), MASH liver disease, and kidney protection in type 2 diabetes with CKD. The evidence for cognitive protection and addiction treatment is promising but not yet at the level of randomized trial confirmation for semaglutide specifically. Emerging weight loss technologies and nutritional approaches may complement pharmacological treatment.
If you're watching this space from a distance, the bigger story may be what GLP-1 research reveals about the connections between metabolic health and diseases we previously considered unrelated. The fact that a drug designed to manage blood sugar can influence heart failure, liver fibrosis, kidney function, brain health, and addiction simultaneously suggests that metabolic dysfunction is a root driver of far more chronic disease than the medical establishment recognized even a decade ago.
The next few years of research will determine which of these early signals hold up under the scrutiny of large randomized trials, and which fade. What's already clear is that the GLP-1 story is about much more than fitting into smaller jeans.
This article discusses current research findings and is not medical advice. Consult your healthcare provider before starting, stopping, or modifying any medication.
Sources
- What's Next for GLP-1s? - Harvard Gazette, February 2026
- The Benefits of GLP-1 Drugs Beyond Weight Loss - Advisory Board, March 2026
- GLP-1 Diabetes Medications Lower Risk of All Kinds of Substance Use Disorders - Scientific American, March 2026
- Weight Loss Drugs and Alzheimer's Disease: Is There Hope for the Future? - Imperial College London, 2026
- Phase 3 Trial of Semaglutide in MASH - New England Journal of Medicine, 2025