For roughly a decade, doctors have had access to a class of drugs that can slash LDL cholesterol (the "bad" kind) by 50 to 60 percent in patients who don't respond well enough to statins alone. These drugs, called PCSK9 inhibitors, work by blocking a protein that destroys LDL receptors on liver cells, letting the liver pull more cholesterol out of the bloodstream. The clinical results have been remarkable. The adoption has not. The reason is simple: PCSK9 inhibitors like evolocumab (Repatha) and alirocumab (Praluent) require injections every two to four weeks. And a large number of patients simply refuse needles, skip doses, or quit treatment entirely. Real-world data show that roughly one in three patients who start injectable PCSK9 therapy discontinue within six months.
That barrier may be about to disappear. A Phase 3 clinical trial published in the New England Journal of Medicine in February 2026 found that enlicitide, an oral PCSK9 inhibitor developed by Merck, reduced LDL cholesterol by up to 60 percent in patients with atherosclerotic cardiovascular disease or at elevated risk. The reduction matched the power of existing injectable therapies, but patients achieved it by swallowing a single pill each morning. If the FDA approves enlicitide, it would become the first oral PCSK9 inhibitor on the market and could fundamentally change how millions of Americans manage their cholesterol.
The CORALreef Lipids Trial: What the Data Show
The trial that produced these results, called CORALreef Lipids, is the largest completed Phase 3 study of enlicitide to date. Led by Ann Marie Navar, M.D., Ph.D., a cardiologist and associate professor at UT Southwestern Medical Center, the study enrolled 2,909 participants across 168 healthcare centers in 14 countries between August 2023 and July 2025. Participants were randomized 2:1 to receive either 20 milligrams of oral enlicitide once daily or a placebo for 52 weeks.
The study population reflected the patients who need this therapy most. Fifty-eight percent had a history of a major atherosclerotic cardiovascular event (heart attack, stroke, or peripheral artery disease) with an LDL level at or above 55 mg/dL. The remaining 42 percent were at risk for a first cardiovascular event with LDL levels at or above 70 mg/dL. All participants were already taking moderate- or high-intensity statins, or had documented statin intolerance. The average age was 63, and 40 percent were women.
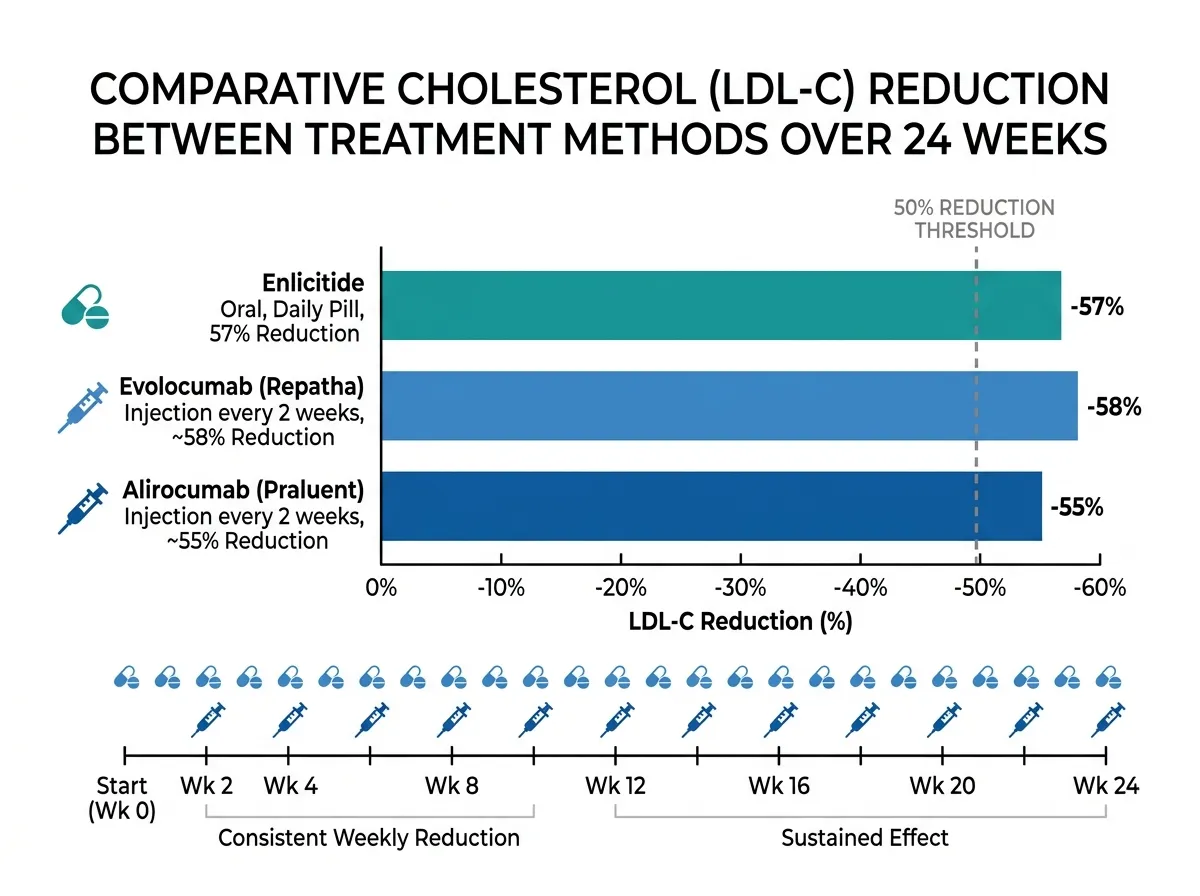
The results were unambiguous. At 24 weeks, participants taking enlicitide saw their LDL cholesterol drop by 57.1 percent compared to baseline, while the placebo group saw a slight 3 percent increase. The adjusted between-group difference was 55.8 percentage points. Seven out of ten patients on enlicitide achieved both a 50 percent or greater LDL reduction and an absolute LDL level below 70 mg/dL, a threshold that current cardiovascular guidelines identify as a treatment goal for high-risk patients.
The benefits extended well beyond LDL. Enlicitide also produced a 53 percent reduction in non-HDL cholesterol (a measure that captures all atherogenic lipoproteins), a 50 percent reduction in apolipoprotein B (the protein component of LDL and other harmful particles), and a 28 percent reduction in lipoprotein(a), a genetically determined risk factor for cardiovascular disease that has historically been extremely difficult to lower with any medication. These reductions were sustained through 52 weeks of treatment, with the between-group LDL difference holding at 47.6 percentage points at the one-year mark.
"Enlicitide is as effective in lowering LDL-C as injectable monoclonal antibody PCSK9 inhibitors," Navar stated after presenting the results at the American Heart Association's Scientific Sessions 2025. She added that "results with enlicitide were numerically better than what has been shown for the siRNA medication inclisiran, which blocks the production of the PCSK9 protein."
Why an Oral PCSK9 Inhibitor Changes Everything
The science behind PCSK9 inhibition is not new. Researchers identified the PCSK9 protein's role in cholesterol metabolism in the early 2000s, and the first injectable PCSK9 inhibitors won FDA approval in 2015. The biology is well understood: PCSK9 binds to LDL receptors on liver cells and marks them for destruction. Fewer LDL receptors means the liver removes less cholesterol from the blood, and LDL levels rise. By blocking PCSK9, these drugs keep more LDL receptors active, allowing the liver to clear cholesterol more efficiently.
The injectable versions have proven their value in large cardiovascular outcomes trials. The FOURIER trial demonstrated that evolocumab reduced the risk of heart attacks and strokes. The ODYSSEY Outcomes trial showed similar results for alirocumab. But the clinical impact of these drugs has been limited by real-world prescribing and adherence patterns. A 2020 analysis published in the Journal of the American Heart Association found that only 29 percent of patients maintained adequate adherence (defined as a proportion of days covered of 80 percent or greater). Nearly 43 percent discontinued therapy within six months. Cost was the most cited barrier, with 40 percent of patients who quit naming price as the primary reason. But injection aversion was a close second, particularly among patients managing multiple chronic conditions who already felt overwhelmed by their treatment regimens.
Enlicitide works through the same biological mechanism but uses a fundamentally different molecular approach. Instead of a large monoclonal antibody that must be injected because the digestive system would destroy it, enlicitide is a macrocyclic peptide, a small, ring-shaped molecule stable enough to survive oral ingestion and absorption through the gut. It binds to PCSK9 and prevents it from interacting with LDL receptors, producing the same downstream effect as the injectable drugs. The CORALreef Lipids trial reported 97 percent adherence with the prescribed dosing regimen, a number that reflects the simplicity of taking a daily pill versus administering a self-injection every two to four weeks.
William E. Boden, M.D., FACC, wrote in an accompanying NEJM editorial that the results provide "compelling evidence that enlicitide provides incremental benefit." He noted that "a simpler formulation of PCSK9 inhibitors that would enable eligible patients to take drugs from this class of medication orally would represent an important step forward to facilitate greater medication adherence and more effective long-term LDL-C lowering for at-risk patients."
The Safety Profile and What Researchers Are Still Watching
One of the most reassuring aspects of the CORALreef Lipids data was the safety profile. Serious adverse events occurred in 10 percent of patients taking enlicitide and 12 percent of patients taking placebo, meaning the drug group actually had a slightly lower rate of serious problems. Discontinuation rates due to adverse events were similar between the two groups. The most common side effects were mild and typical of large clinical trials: upper respiratory infections, COVID-19, and musculoskeletal pain, none of which occurred at meaningfully higher rates in the enlicitide group than in the placebo group.
This matters because safety concerns have historically slowed the adoption of cholesterol-lowering drugs. When statins first became widely prescribed, fears about muscle pain, liver damage, and cognitive effects (most of which turned out to be overstated or rare) dampened patient willingness to start therapy. The early PCSK9 inhibitor trials also drew scrutiny over potential neurocognitive effects, though subsequent long-term data have been largely reassuring. Enlicitide's clean safety record through 52 weeks of treatment removes one potential objection that could slow its path to widespread use.

That said, 52 weeks is not the same as five or ten years. The long-term safety of enlicitide remains an open question, as it does for any new medication class. Merck has already enrolled more than 14,500 participants in a larger cardiovascular outcomes trial called CORALreef Outcomes, which will evaluate whether enlicitide's cholesterol-lowering effects translate into fewer heart attacks, strokes, and cardiovascular deaths over a longer follow-up period. This is the same type of outcomes trial that cemented the injectable PCSK9 inhibitors as proven cardiovascular therapies, and it will be essential for establishing enlicitide's full clinical value. Additional CORALreef data are scheduled for presentation at ACC.26 (the American College of Cardiology annual meeting) in New Orleans on March 28-30, 2026.
Navar acknowledged the clinical reality: "There are a number of patients who would prefer an oral to an injectable therapy, and I do have patients who will not initiate an injection and only want to take an oral medication." The CORALreef Outcomes trial will determine whether giving those patients an oral option translates into better cardiovascular outcomes at the population level.
Where Enlicitide Fits Among Current Treatments
Cholesterol management in 2026 involves a tiered approach. Statins remain the first-line therapy for most patients, and they work well for a majority. But roughly 25 to 30 percent of patients on maximum-tolerated statin therapy still cannot reach their LDL targets, particularly those with established cardiovascular disease who need to push LDL below 55 or 70 mg/dL. Another subset of patients cannot tolerate statins at all due to muscle symptoms. These are the populations that stand to benefit most from enlicitide.
The current add-on options include ezetimibe (a daily pill that blocks cholesterol absorption in the gut, reducing LDL by roughly 20 percent), bempedoic acid (another oral option that targets cholesterol production through a different pathway than statins, lowering LDL by about 18 percent), the injectable PCSK9 antibodies (50 to 60 percent LDL reduction), and inclisiran (a twice-yearly injection that blocks PCSK9 production, reducing LDL by roughly 50 percent). Enlicitide would slot into this hierarchy as a once-daily oral option with the potency of injectables, a combination that currently does not exist.
The evolving pharmaceutical market in metabolic and cardiovascular medicine has shown that route of administration can dramatically affect how widely a drug gets used. GLP-1 receptor agonists saw their adoption explode when oral semaglutide became available alongside the injectable version. The same dynamic could play out for PCSK9 inhibitors. A cardiologist or primary care physician who hesitates to prescribe a biweekly injection (with its prior authorization requirements, cold chain storage, and patient training) might readily prescribe a daily pill.
The lipoprotein(a) reduction is worth noting separately. Lipoprotein(a), or Lp(a), is an inherited risk factor that affects roughly 20 percent of the global population at elevated levels. Unlike LDL cholesterol, Lp(a) does not respond to statins, exercise, or dietary changes. Until recently, no approved therapy could meaningfully lower it. Enlicitide's 28 percent reduction in Lp(a) is not as dramatic as the reductions seen with experimental antisense therapies specifically targeting Lp(a), but it is a meaningful secondary benefit for patients who happen to carry elevated levels. For someone already taking enlicitide for LDL reduction who also has high Lp(a), the dual benefit could be particularly significant.
What This Could Mean for Millions
Heart disease remains the leading cause of death in the United States, killing roughly 700,000 Americans each year. Elevated LDL cholesterol is one of the most modifiable risk factors, and the relationship between lower LDL and fewer cardiovascular events is among the most well-established in all of medicine. Every 39 mg/dL reduction in LDL cholesterol corresponds to approximately a 22 percent decrease in major cardiovascular events, according to a meta-analysis published in The Lancet. For patients who achieve the 55 to 60 percent reductions seen with enlicitide, the absolute risk reduction could be substantial.
The practical promise of enlicitide comes down to a simple calculation. If a drug works as well as existing injectables but more patients are willing to take it (and keep taking it), the population-level benefit could far exceed what the injectable PCSK9 inhibitors have delivered despite being available for over a decade. The 97 percent adherence rate observed in the CORALreef trial, while measured in the controlled setting of a clinical study, hints at what might be possible when you remove the injection barrier. Even if real-world adherence proves lower, an oral option that retains even 70 to 80 percent of patients at one year would be a substantial improvement over the roughly 57 percent retention rate observed with injectable PCSK9 therapies.
Enlicitide has not yet received FDA approval, and timing for a regulatory submission has not been publicly announced. The ongoing CORALreef Outcomes trial, with its 14,500-plus participants, will provide the cardiovascular event data that regulators and clinicians typically want to see before granting broad prescribing recommendations. Merck also received an FDA National Priority Review voucher for enlicitide, signaling the agency's recognition that the drug addresses a significant unmet medical need. For patients currently living with elevated cardiovascular risk who have not been able to reach their cholesterol targets, the possibility of achieving injectable-level results with a morning pill is the most significant development in lipid management in years.
The bottom line for patients: if you're on a statin and your LDL cholesterol is still above your target, talk to your cardiologist or primary care doctor about your options. Ask specifically whether a PCSK9 inhibitor might be appropriate for your risk profile. And if the idea of self-injecting every two weeks has kept you from exploring that option, enlicitide may soon offer an alternative that makes the conversation much simpler.
This article discusses current research findings and is not medical advice. Consult your healthcare provider before starting, stopping, or modifying any medication.
Sources
- Navar AM, et al. "A Placebo-Controlled Trial of the Oral PCSK9 Inhibitor Enlicitide." New England Journal of Medicine, February 4, 2026. https://www.nejm.org/doi/full/10.1056/NEJMoa2511002
- Merck. "Merck's Enlicitide Decanoate, an Investigational Oral PCSK9 Inhibitor, Significantly Reduced LDL-C in Phase 3 CORALreef Lipids Trial." Press Release. https://www.merck.com/news/mercks-enlicitide-decanoate-an-investigational-oral-pcsk9-inhibitor-significantly-reduced-ldl-c-in-phase-3-coralreef-lipids-trial/
- American Heart Association. "Investigational daily pill lowered bad cholesterol as much as injectables." November 2025. https://newsroom.heart.org/news/investigational-daily-pill-lowered-bad-cholesterol-as-much-as-injectables
- American College of Cardiology. "CORALreef Lipids: Oral PCSK9 Inhibitor Enlicitide Reduces LDL-C in High-Risk Patients." Journal Scan, February 2026. https://www.acc.org/latest-in-cardiology/journal-scans/2026/02/09/18/56/coralreef-lipids
- UT Southwestern Medical Center. "Experimental pill dramatically reduces 'bad' cholesterol." February 2026. https://www.utsouthwestern.edu/newsroom/articles/year-2026/feb-experimental-pill-bad-cholesterol.html